Potentiation of adipogenesis and insulinomimetic effects of a novel vanadium complex (N′-[(E)-(5-bromo-2-oxophenyl) methylidene]-4-methoxybenzohydrazide)oxido- (1,10-phenanthroline)vanadium(IV)in 3T3-L1 cells
DOI:
https://doi.org/10.5604/01.3001.0013.1048Keywords:
vanadium complexes, phosphatases inhibition, adipocytes, adipogenesis, glucose utilisationAbstract
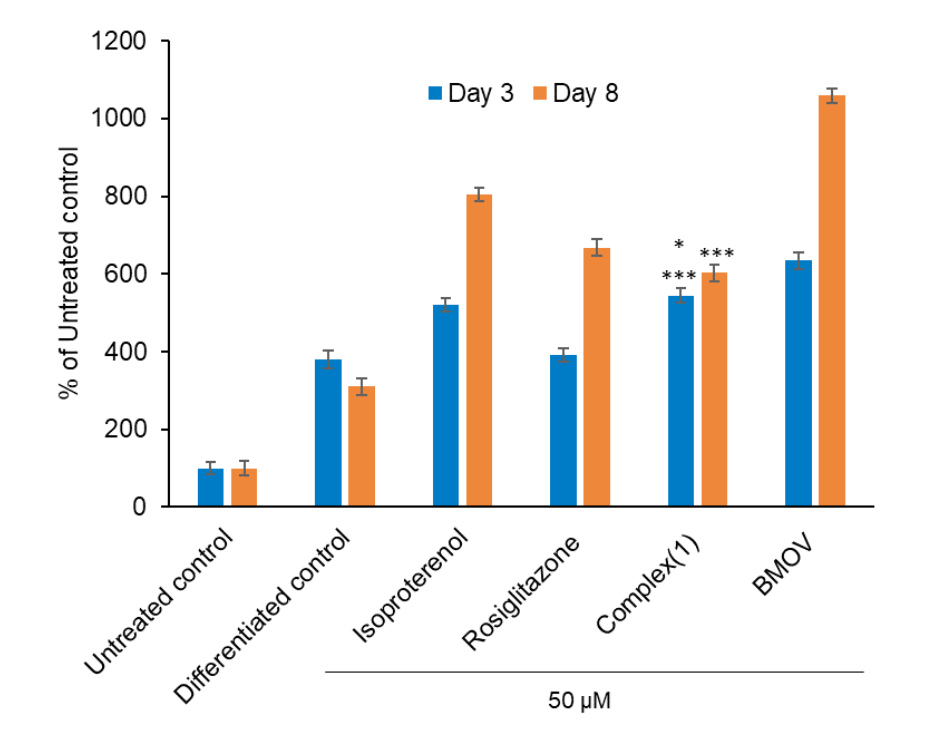
The latest results in action of vanadium compounds show important influence on adipogenesis processes and adipocytes function. Based on previously screening tests in cellular models the novel vanadium complex (N'-[(E)-(5-bromo-2-oxophenyl)methylidene]-4-methoxybenzohydrazide)oxido(1,10-phenanthroline)vanadium(IV) was selected for this study. This complex shown potent inhibition ability of tyrosine phosphatases and differences in the degree of inhibition particular phosphatases was observed. Significantly increasing of intracellular lipid accumulation and proliferative effect on 3T3-L1 preadipocytes confirmed the ability of this complex to enhancement of adipogenesis. The insulinomimetic activity of the tested complex was also demonstrated in fully differentiated 3T3-L1 adipocytes, in which glucose utilization was potentiated. Presented results support the thesis that vanadium complexes show promising possibilities for opportunities of new therapeutic strategies for the treatment of type 2 diabetes involved adipocytes.
Downloads
References
Rosen ED, Spiegelman BM. Adipocytes as regulators of energy balance and glucose homeostasis. Nature. 2006; 14;444(7121):847-853. Google Scholar
Simha V, Garg A. Lipodystrophy: lessons in lipid and energy metabolism. Curr Opin Lipidol. 2006; 17(2):162-169. Google Scholar
Neeland IJ, Turer AT, Ayers CR, Powell-Wiley TM, Vega GL, Farzaneh-Far R, Grundy SM, Khera A, McGuire DK, de Lemos JA. Dysfunctional adiposity and the risk of prediabetes and type 2 diabetes in obese adults. JAMA. 2012; 308(11):1150-1159 Google Scholar
Lefterova MI, Lazar MA. New developments in adipogenesis. Trends Endocrinol Metab. 2009; 20(3):107-114. Google Scholar
Farmer SR. Transcriptional control of adipocyte formation. Cell Metab. 2006; 4(4):263-273. Google Scholar
Rosen, ED, Spiegelman, BM. Molecular regulation of adipogenesis. Annual Review of Cell Biology, 2000; 16, 145-171. Google Scholar
Mota de Sá P, Richard AJ, Hang H, Stephens JM. Transcriptional Regulation of Adipogenesis. Compr Physiol. 2017; (2):635-674. Google Scholar
Lefterova MI, Lazar MA. New developments in adipogenesis. Trends Endocrinol Metab. 2009; 20(3):107-114. Google Scholar
Chaudhary S, Dube A, Kothari V, Sachan N, Upasani CD. NS-1: a novel partial peroxisome proliferator-activated receptor γ agonist to improve insulin sensitivity and metabolic profile. Eur J Pharmacol. 2012; 684(1-3):154-160. Google Scholar
Mayer P, Haas B, Celner J, Enzmann H, Pfeifer A. Glitazone-like action of glimepiride and glibenclamide in primary human adipocytes. Diabetes Obes Metab. 2011; 13(9):791-799. Google Scholar
Haas B, Schlinkert P, Mayer P, Eckstein N. Targeting adipose tissue. Diabetol Metab Syndr. 2012; 4(1):43. Google Scholar
Dubyak GR, Kleinzeller A. The insulin-mimetic effects of vanadate in isolated rat adipocytes. Dissociation from effects of vanadate as a (Na+-K+)ATPase inhibitor. J Biol Chem. 1980; 255(11):5306-5312. Google Scholar
Goldstein BJ. Regulation of insulin receptor signaling by protein-tyrosine dephosphorylation. Receptor. 1993; 3(1):1-15. Google Scholar
Bae KH, Kim WK, Lee SC. Involvement of protein tyrosine phosphatases in adipogenesis: new anti-obesity targets? BMB Rep. 2012; 45(12):700-706. Google Scholar
Irving E, Stoker AW. Vanadium Compounds as PTP Inhibitors. Molecules. 2017; 22(12): E2269. Google Scholar
Green H, Meuth M. An established pre-adipose cell line and its differentiation in culture. Cell. 1974; 3(2):127-133. Google Scholar
Hiromura M, Nakayama A, Adachi Y, Doi M, Sakurai H. Action mechanism of bis(allixinato)oxovanadium(IV) as a novel potent insulin-mimetic complex: regulation of GLUT4 translocation and FoxO1 transcription factor. J Biol Inorg Chem. 2007; 12(8):1275-1287. Google Scholar
Zuo YQ, Liu WP, Niu YF, Tian CF, Xie MJ, Chen XZ, Li L. Bis(alpha-furancarboxylato)oxovanadium(IV) prevents and improves dexamethasone-induced insulin resistance in 3T3-L1 adipocytes. J Pharm Pharmacol. 2008; 60(10):1335-1340. Google Scholar
Liu JC, Yu Y, Wang G, Wang K, Yang XG. Bis(acetylacetonato)-oxovanadium(iv), bis(maltolato)-oxovanadium(iv) and sodium metavanadate induce antilipolytic effects by regulating hormone-sensitive lipase and perilipin via activation of Akt. Metallomics. 2013; 5(7):813-820. Google Scholar
Seale AP, de Jesus LA, Park MC, Kim YS. Vanadium and insulin increase adiponectin production in 3T3-L1 adipocytes. Pharmacol Res. 2006; 54(1):30-38. Google Scholar
Halevas E, Tsave O, Yavropoulou MP, Hatzidimitriou A, Yovos JG, Psycharis V, Gabriel C, Salifoglou A. Design, synthesis and characterization of novel binary V(V)-Schiff base materials linked with insulin-mimetic vanadium-induced differentiation of 3T3-L1 fibroblasts to adipocytes. Structure-function correlations at the molecular level. J Inorg Biochem. 2015; 147:99-115. Google Scholar
Zhang L, Huang Y, Liu F, Zhang F, Ding W. Vanadium(IV)-chlorodipicolinate inhibits 3T3-L1 preadipocyte adipogenesis by activating LKB1/AMPK signaling pathway. J Inorg Biochem. 2016; 162:1-8. Google Scholar
Kazek G, Głuch-Lutwin M, Mordyl B, Menaszek E, Szklarzewicz J, Gryboś R, PapieżM. Cell-based Screening For Identification Of The Novel Vanadium Complexes with Multidirectional Activity Relative To The Cells And The Mechanisms Associated With Metabolic Disorders. Sci. Tech., Innov. 2019; 4(1):47-54. Google Scholar
Zabierowski P, Szklarzewicz J, Gryboś R, Modryl B, Nitek W. Assemblies of salen-type oxidovanadium(IV) complexes: substituent effects and in vitro protein tyrosine phosphatase inhibition. Dalton Trans. 2014; 43(45):17044-17053. Google Scholar
Welte S, Baringhaus KH, Schmider W, Müller G, Petry S, Tennagels N. 6,8-Difluoro-4-methylumbiliferyl phosphate:a fluorogenic substrate for protein tyrosine phosphatases. Anal Biochem. 2005; 338(1):32-38. Google Scholar
Poulos SP, Dodson MV, Hausman GJ. Cell line models for differentiation: preadipocytes and adipocytes. Exp Biol Med. (Maywood). 2010; 235(10):1185-1193. Google Scholar
Green H, Kehinde O. An established preadipose cell line and its differentiation in culture. II. Factors affecting the adipose conversion. Cell. 1975; 5(1):19-27. Google Scholar
Thompson KH, Lichter J, LeBel C, Scaife MC, McNeill JH, Orvig C. Vanadium treatment of type 2 diabetes: a view to the future. J Inorg Biochem. 2009; 103(4):554-558. Google Scholar
Smith U, Kahn BB. Adipose tissue regulates insulin sensitivity: role of adipogenesis, de novo lipogenesis and novel lipids. J Intern Med. 2016; 280(5):465-475. Google Scholar
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2019 University of Applied Sciences in Tarnow, Poland & Authors

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.



